Challenge:
A team of scientists at a large pharmaceutical company were spending a lot of time coordinating Quality Control (QC) and shipping logistics, which was not a core competency or activity that would drive their pipeline forward. Additionally, storage of produced reagents was beginning to take up too much space in their on-site freezers.
Details:
The scientists were ready to hand off activity that began as a minimal time commitment, but morphed into a larger part of their routine as the workflow became a requirement for more of their pipeline programs.
The challenge was finding a provider that could take on the workflow from end to end, to avoid coordinating different steps in the workflow with multiple partners. This required a supplier that could do cell culture, QC their reagents via flow cytometry, store the QC’d material, and easily handle the logistics of shipping the materials to the client’s Contract Research Organizations (CROs) upon request and within an acceptable time-frame.
Expertise in cell culture, biospecimen storage, 30 plus years’ experience with coordinating shipments both within the United States and internationally, and access to specialized flow cytometry made ABS a perfect fit!
The ABS team collaborated with the client to gain a deep understanding of their current situation and ultimate goals for outsourcing this activity. Together they designed a custom solution specific to their needs.
Solution: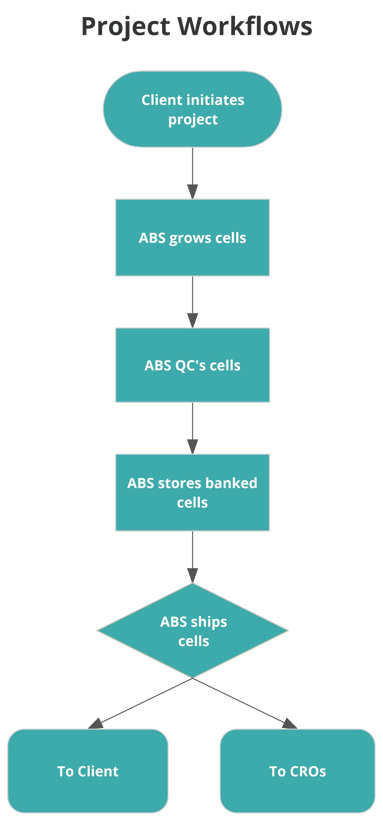
ABS created a custom workflow with the client that allows them to trigger the newly outsourced process with a simple email. The client completes a request template designed specifically for this workflow each time the process needs to be triggered. The completed template is attached to the email and the process is initiated. The requestor then receives a confirmation email upon completion.
Now ABS handles the following:
- Coordination with applicable CROs involved
- Shipments to and from those CROs as needed
- Stores and maintains the inventory of the client’s reagents
- Sends alerts to the client when their inventory drops below a certain threshold
- Provides automated inventory status reports monthly
ABS continues to meet with the client quarterly to conduct a business review, where workflow changes or improvements are discussed, as well as plans for the upcoming quarter to ensure the continued success of this project.
Benefits:
The tailored solution provided by ABS reduced the number of touch-points required to complete the workflow, increasing process efficiency. ABS’ storage facility freed up more on-site capacity for reagents that need to be readily available.
Now the scientists who used to spend time on these important “non-core” activities are free to concentrate their efforts on the critical scientific discovery that drives their pipeline programs toward successful completion.
Additionally, with over 30 years of shipping experience, the client can trust that the experts at ABS will handle the shipping process and logistics coordination of critical reagents and other regulated goods.
Please contact ABS for more information:
(302) 6544492
(800) 391-2391





